The prevalence of mutualism nature, in which different species cooperate for common benefit, is difficult to explain. Theory conflicts on whether mutualisms should be stable or abandoned over evolutionary time. On the one hand, natural selection is expected to favour exploitation over cooperation because individuals that derive benefit from a mutualistic partner can maximize their own fitness by cheating, or otherwise avoiding the costs of cooperation. On the other hand, a mutualism can persist if there is natural selection for cooperation, or if mutations that reduce cooperation are rare.
One of the most common mutualisms in nature is the symbiosis between plants and root inhabiting mycorrhizal fungi. Plants provide the fungi with sugars from photosynthesis and in return, the fungi assist plants in accessing hard to acquire nutrients in soil. Because plants gain nutritional benefits from the fungi, the symbiosis is important for plant productivity in both natural and agricultural ecosystems. Mycorrhizal symbioses are dominated by plant interactions with arbuscular mycorrhizal (AM) fungi (~70% of all species), with a minority (<5%) engaged in an alternate symbiosis occurs with ectomycorrhizal (EM) fungi. About 10-15% of plant species are able to grow either with or without AM fungi, and are considered to be ‘weak’ AM mutualists. An additional 10% of species have lost the capacity to form a symbiosis and are categorized as non-mycorrhizal. As with mutualisms in general, explaining the evolutionary persistence of the mycorrhizal symbiosis is a long-standing question in biology.
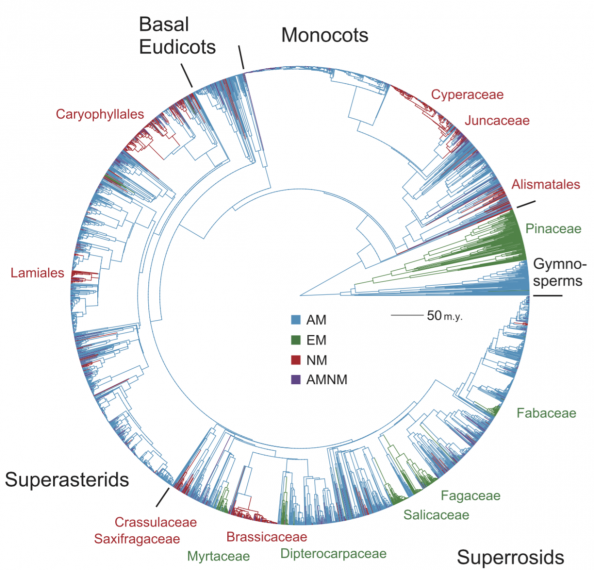
To understand why the mycorrhizal symbiosis has persisted over >400 million years of evolution, a team of researchers from Canada, the USA and Australia, led by Dr. Hafiz Maherali, reconstructed the evolution of the symbiosis using a phylogeny (a genetic record of shared ancestry) and information on the mycorrhizal status of plant species [1]. The reconstruction showed that the common ancestor of seed plants most likely associated with AM fungi and that the EM, weak AM and non-mycorrhizal states descended from the AM state. Direct transitions among the primary AM, EM and non-mycorrhizal states were infrequent and generally irreversible, implying that natural selection or a lack of genetic variation could promote stasis once a particular state evolved. The researchers also found that the weak AM state was a necessary precursor to the evolution of complete mutualism abandonment (i.e., the non-mycorrhizal state). Nevertheless, lineages of plants were much more likely to revert back to the AM state from the weak AM state rather than proceed to mutualism abandonment.
These findings suggest that the mycorrhizal symbiosis persists because natural selection for cooperation is more common than natural selection for mutualism abandonment, and that cheating within a mutualism may be rarer than previously thought. By identifying how likely it is that the symbiosis will be retained or lost in different groups of plants, the work will assist in planning future experiments that probe for specific causes of mutualism persistence and abandonment. Identifying these causes will assist in predicting how anthropogenic activities like climate change and fertilizer deposition will affect the mycorrhizal mutualism, and what conservation efforts are required for its maintenance.
